AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Thus, it takes much more energy than just overcoming a larger ionic charge would suggest. And the element which has the lowest ionization energy is Caesium in 3.8939 eV. The first ionization energy refers to the energy it takes to remove one electron from an atom. The element which has the highest ionization energy is Helium with 24.58741 eV. P A: (a) First ionization energy of P > first ionization energy of I. which element of the following pairs has the higher first ionization energy and can you show me how you know O vs. Between Mg and Ca, the element with the higher first ionization energy is - 3. You can print the list of elements by hitting the print button below. Q: Choose the element with the higher first ionization energy in each pair.a. Between Kr and Br, the element with the higher first ionization energy is - 2. Why is it so much larger? Because the first two electrons are removed from the 3 s subshell, but the third electron has to be removed from the n = 2 shell (specifically, the 2 p subshell, which is lower in energy than the n = 3 shell). To list the elements order by ionization energy, click on the table headers. Based on trends in ionization energies across a row and down a column, identify the. Strategy: Locate the elements in the periodic table. Asked for: element with lowest first ionization energy. The first ionization energy for an element, X, is the energy required to form a cation with +1 charge: X(g) X+(g) +e IE1 (3.3.1) (3.3.1) X ( g) X + ( g) + e IE 1.  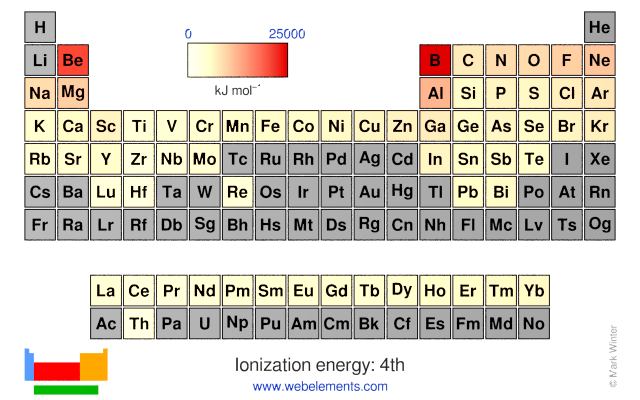
The third IE, however, is over five times the previous one. Use their locations in the periodic table to predict which element has the lowest first ionization energy: Ca, K, Mg, Na, Rb, or Sr. 
Ca (Calcium) will have high ionization energy. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion. Question: Which of the following elements has the smallest first ionization energy A) S B) O C) Si D) Sr E) Cs.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
